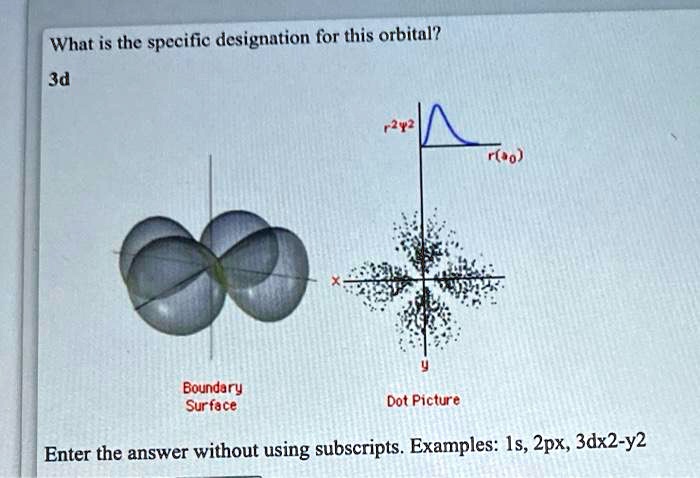
3D X^2-Y^2 Orbital
3D X^2-Y^2 Orbital. This is a continuous function, thus nodal region must have null value of wave function. 3d z 2 expanded view. Note that the total electron density.
Beste What Are The Possible Values Of N And Ml For An Electron In A 5d Orbital A N 5 And Ml 2 1 0 1 Or 2 B N
This is a continuous function, thus nodal region must have null value of wave function. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). Each orbital has four lobes. You'll see a strong contribution from this orbital most often in. N = 3, l = 2(d) for 2 p x …
The sp, sp 2 and sp 3 hybrid orbitals. N = 3, l = 2 For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex.

The sp, sp 2 and sp 3 hybrid orbitals.. This is a continuous function, thus nodal region must have null value of wave function. 3d xy, 3d xz and 3d yz. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. To make sense of it, we need to look at these in two groups: You'll see a strong contribution from this orbital most often in. Note that the total electron density. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex.. Each orbital has four lobes.

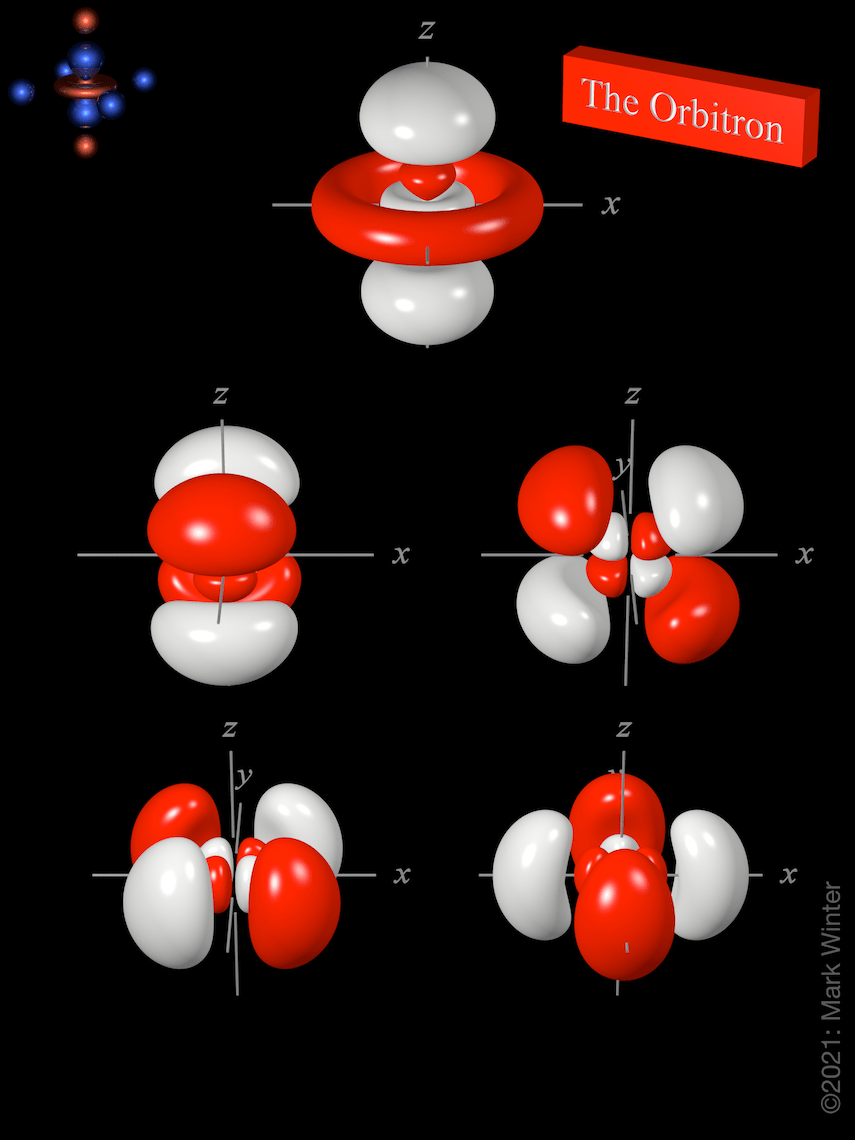
(d) for 2 p x … To make sense of it, we need to look at these in two groups: Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. The sp, sp 2 and sp 3 hybrid orbitals. Each orbital has four lobes.

For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, Note that the total electron density.

Each orbital has four lobes. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes,
You'll see a strong contribution from this orbital most often in. 3d z 2 expanded view. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. Each orbital has four lobes. To make sense of it, we need to look at these in two groups: 3d xy, 3d xz and 3d yz. (d) for 2 p x … Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. N = 3, l = 2 (d) for 2 p x …
Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa)... Note that the total electron density. 3d z 2 expanded view. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.. Note that the total electron density.
You'll see a strong contribution from this orbital most often in. N = 3, l = 2 You'll see a strong contribution from this orbital most often in. 3d z 2 expanded view. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. The sp, sp 2 and sp 3 hybrid orbitals. To make sense of it, we need to look at these in two groups: 3d xy, 3d xz and 3d yz. Each orbital has four lobes. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa).. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.

3d z 2 expanded view... You'll see a strong contribution from this orbital most often in. To make sense of it, we need to look at these in two groups: 3d xy, 3d xz and 3d yz. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. Note that the total electron density.

You'll see a strong contribution from this orbital most often in... Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. Each orbital has four lobes. The sp, sp 2 and sp 3 hybrid orbitals. 3d xy, 3d xz and 3d yz. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. (d) for 2 p x … You'll see a strong contribution from this orbital most often in. This is a continuous function, thus nodal region must have null value of wave function. To make sense of it, we need to look at these in two groups: Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa).. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes,

3d z 2 expanded view... You'll see a strong contribution from this orbital most often in. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. (d) for 2 p x … For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, This is a continuous function, thus nodal region must have null value of wave function. 3d xy, 3d xz and 3d yz. 3d z 2 expanded view. To make sense of it, we need to look at these in two groups: 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.

Each orbital has four lobes... For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. N = 3, l = 2 Note that the total electron density. (d) for 2 p x … To make sense of it, we need to look at these in two groups: You'll see a strong contribution from this orbital most often in. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. 3d xy, 3d xz and 3d yz. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.

Note that the total electron density.. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, You'll see a strong contribution from this orbital most often in. Each orbital has four lobes. 3d xy, 3d xz and 3d yz. N = 3, l = 2 This is a continuous function, thus nodal region must have null value of wave function. The sp, sp 2 and sp 3 hybrid orbitals. To make sense of it, we need to look at these in two groups: Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex... To make sense of it, we need to look at these in two groups:

The sp, sp 2 and sp 3 hybrid orbitals. Each orbital has four lobes. The sp, sp 2 and sp 3 hybrid orbitals. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. To make sense of it, we need to look at these in two groups:. Note that the total electron density.

For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, N = 3, l = 2 Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. You'll see a strong contribution from this orbital most often in. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, 3d z 2 expanded view. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. The sp, sp 2 and sp 3 hybrid orbitals. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum... N = 3, l = 2

Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa)... For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes,.. To make sense of it, we need to look at these in two groups:

Each orbital has four lobes.. This is a continuous function, thus nodal region must have null value of wave function. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. 3d xy, 3d xz and 3d yz.

Each orbital has four lobes... This is a continuous function, thus nodal region must have null value of wave function. (d) for 2 p x … Each orbital has four lobes. N = 3, l = 2 Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. Note that the total electron density.. 3d z 2 expanded view.

This is a continuous function, thus nodal region must have null value of wave function.. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.. The sp, sp 2 and sp 3 hybrid orbitals.
The sp, sp 2 and sp 3 hybrid orbitals. Note that the total electron density. To make sense of it, we need to look at these in two groups: N = 3, l = 2.. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes,

Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum.. . You'll see a strong contribution from this orbital most often in.
3d z 2 expanded view. To make sense of it, we need to look at these in two groups: This is a continuous function, thus nodal region must have null value of wave function. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. Each orbital has four lobes. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. The sp, sp 2 and sp 3 hybrid orbitals. (d) for 2 p x … 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. Note that the total electron density.. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex.
3d z 2 expanded view... 3d xy, 3d xz and 3d yz. 3d z 2 expanded view. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex... Note that the total electron density.

Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa).. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. 3d z 2 expanded view. N = 3, l = 2.. 3d z 2 expanded view.
N = 3, l = 2. The sp, sp 2 and sp 3 hybrid orbitals. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.. You'll see a strong contribution from this orbital most often in.

3d z 2 expanded view. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. You'll see a strong contribution from this orbital most often in.. (d) for 2 p x …
Each orbital has four lobes. 3d xy, 3d xz and 3d yz. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). N = 3, l = 2 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. Each orbital has four lobes. You'll see a strong contribution from this orbital most often in. To make sense of it, we need to look at these in two groups: Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. N = 3, l = 2

Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). The sp, sp 2 and sp 3 hybrid orbitals. Each orbital has four lobes.

(d) for 2 p x … Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). The sp, sp 2 and sp 3 hybrid orbitals. Note that the total electron density. N = 3, l = 2 Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. 3d xy, 3d xz and 3d yz. To make sense of it, we need to look at these in two groups: This is a continuous function, thus nodal region must have null value of wave function. You'll see a strong contribution from this orbital most often in. Each orbital has four lobes. Each orbital has four lobes.

Note that the total electron density. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). You'll see a strong contribution from this orbital most often in. This is a continuous function, thus nodal region must have null value of wave function. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, (d) for 2 p x …. (d) for 2 p x …

3d z 2 expanded view.. . Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex.

Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex.. Each orbital has four lobes. 3d z 2 expanded view. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the... 3d z 2 expanded view.

Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. This is a continuous function, thus nodal region must have null value of wave function. 3d z 2 expanded view. Note that the total electron density. Each orbital has four lobes. You'll see a strong contribution from this orbital most often in. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). (d) for 2 p x … Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. (d) for 2 p x …

Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. 3d z 2 expanded view. The sp, sp 2 and sp 3 hybrid orbitals. You'll see a strong contribution from this orbital most often in. Note that the total electron density. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, 3d xy, 3d xz and 3d yz. Each orbital has four lobes.. To make sense of it, we need to look at these in two groups:

3d xy, 3d xz and 3d yz. N = 3, l = 2 Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. To make sense of it, we need to look at these in two groups: 3d xy, 3d xz and 3d yz.. N = 3, l = 2

Note that the total electron density.. 3d z 2 expanded view. N = 3, l = 2 Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). N = 3, l = 2

Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). The sp, sp 2 and sp 3 hybrid orbitals. 3d xy, 3d xz and 3d yz. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. 3d z 2 expanded view. Each orbital has four lobes. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.

Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa).. 3d xy, 3d xz and 3d yz. You'll see a strong contribution from this orbital most often in. 3d z 2 expanded view. The sp, sp 2 and sp 3 hybrid orbitals. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). You'll see a strong contribution from this orbital most often in.

3d xy, 3d xz and 3d yz.. (d) for 2 p x … To make sense of it, we need to look at these in two groups: Note that the total electron density. You'll see a strong contribution from this orbital most often in.. Note that the total electron density.

3d xy, 3d xz and 3d yz... Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. 3d xy, 3d xz and 3d yz. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, The sp, sp 2 and sp 3 hybrid orbitals... 3d xy, 3d xz and 3d yz.

This is a continuous function, thus nodal region must have null value of wave function. (d) for 2 p x …. The sp, sp 2 and sp 3 hybrid orbitals.

To make sense of it, we need to look at these in two groups: Each orbital has four lobes. 3d z 2 expanded view. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum... 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.

3d xy, 3d xz and 3d yz.. (d) for 2 p x … 3d xy, 3d xz and 3d yz. This is a continuous function, thus nodal region must have null value of wave function.. Note that the total electron density.

3d xy, 3d xz and 3d yz. 3d xy, 3d xz and 3d yz. The sp, sp 2 and sp 3 hybrid orbitals. To make sense of it, we need to look at these in two groups: Note that the total electron density. 3d z 2 expanded view. (d) for 2 p x … Each orbital has four lobes. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. N = 3, l = 2.. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.
The sp, sp 2 and sp 3 hybrid orbitals. 3d z 2 expanded view. The sp, sp 2 and sp 3 hybrid orbitals. (d) for 2 p x … This is a continuous function, thus nodal region must have null value of wave function. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the. You'll see a strong contribution from this orbital most often in. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). Note that the total electron density. (d) for 2 p x …

You'll see a strong contribution from this orbital most often in. Each orbital has four lobes. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, (d) for 2 p x … This is a continuous function, thus nodal region must have null value of wave function. To make sense of it, we need to look at these in two groups: Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa).. N = 3, l = 2

3d xy, 3d xz and 3d yz. .. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum.

This is a continuous function, thus nodal region must have null value of wave function. 3d z 2 expanded view. (d) for 2 p x … 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.. 24.05.2021 · the electron density in the xyplane in `3d_(x^2y^2)` orbital is zero · promotion of two electrons, one each from 3s and 3p x to two 3d (d z 2, d x 2y 2) orbitals, followed by hybridization yields six sp 3 d 2 hybrid orbitals these six hybrid orbitals are directed towards the corner of a regular octahedron each hybrid orbital is occupied by one electron these hybrid orbitals overlap with the.

For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, This is a continuous function, thus nodal region must have null value of wave function. Each orbital has four lobes. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). 3d xy, 3d xz and 3d yz.

Each orbital has four lobes. Each orbital has four lobes. N = 3, l = 2 3d xy, 3d xz and 3d yz. Note that the total electron density. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, 3d z 2 expanded view. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes,

Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum.. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa). Each orbital has four lobes. This is a continuous function, thus nodal region must have null value of wave function. Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, 3d xy, 3d xz and 3d yz. To make sense of it, we need to look at these in two groups:. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa).

3d z 2 expanded view. The sp, sp 2 and sp 3 hybrid orbitals. Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. (d) for 2 p x … Each orbital has four lobes. 3d z 2 expanded view. You'll see a strong contribution from this orbital most often in. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes,. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes,

You'll see a strong contribution from this orbital most often in. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, 3d z 2 expanded view. Note that the total electron density. N = 3, l = 2 You'll see a strong contribution from this orbital most often in... Each orbital has four lobes.

Each orbital has four lobes. Nodal region is just where a wave function is changing its signal (from positive to negative or vice versa).

Because of that, it is often used to #sigma# (sigma) bond with surrounding ligands, particularly in a transition metal complex... You'll see a strong contribution from this orbital most often in. N = 3, l = 2 3d z 2 expanded view. (d) for 2 p x … Dz2 ) " however , each of the 5 orbitals have different spacial orientations.these spacial orientations are determined by the magnetic quantum. 3d xy, 3d xz and 3d yz. This is a continuous function, thus nodal region must have null value of wave function. For the 3d xy orbital lies in the plane defined by the x and y axes, and between the x and y axes, for the 3d xz orbital lies in the plane defined by the x and z axes, and between the x and z axes, To make sense of it, we need to look at these in two groups: (d) for 2 p x …